Amorphous Solid Dispersions
Visualize and control particle microstructure to improve bioavailability, stability, and manufacturability at every stage of development.

Who this is for
Drug product and process development teams working on spray‑dried dispersions (SDDs) for tablets, capsules, granules, and DPIs, especially when scaling across sites or CDMOs.
The Problem
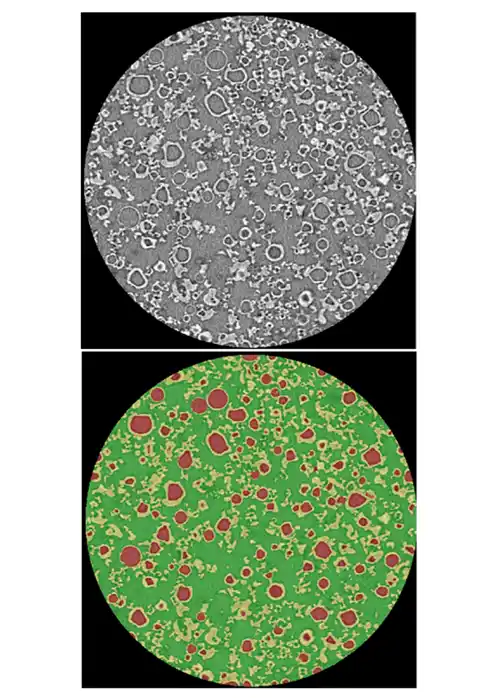
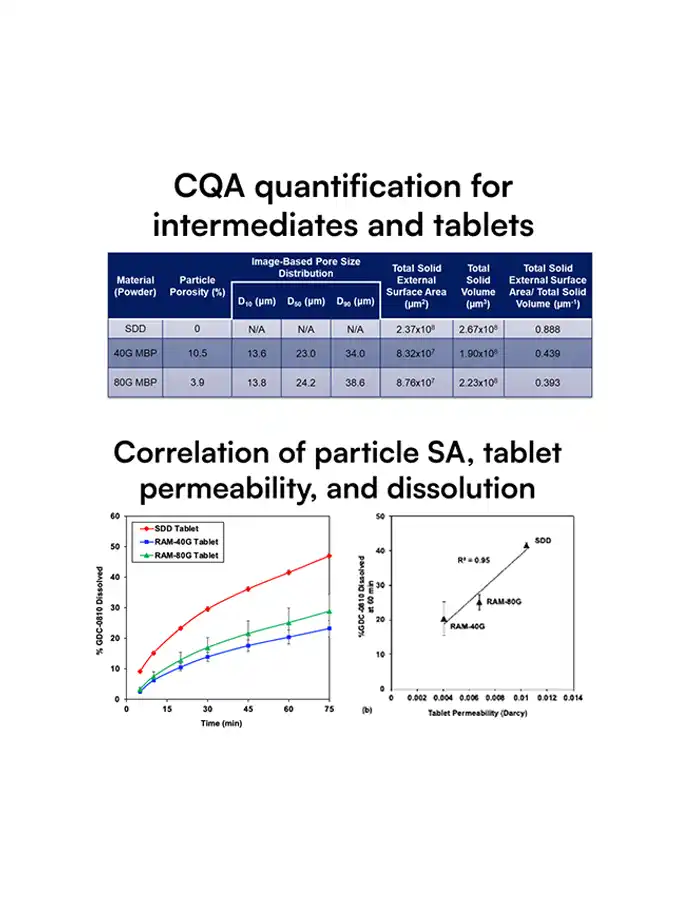
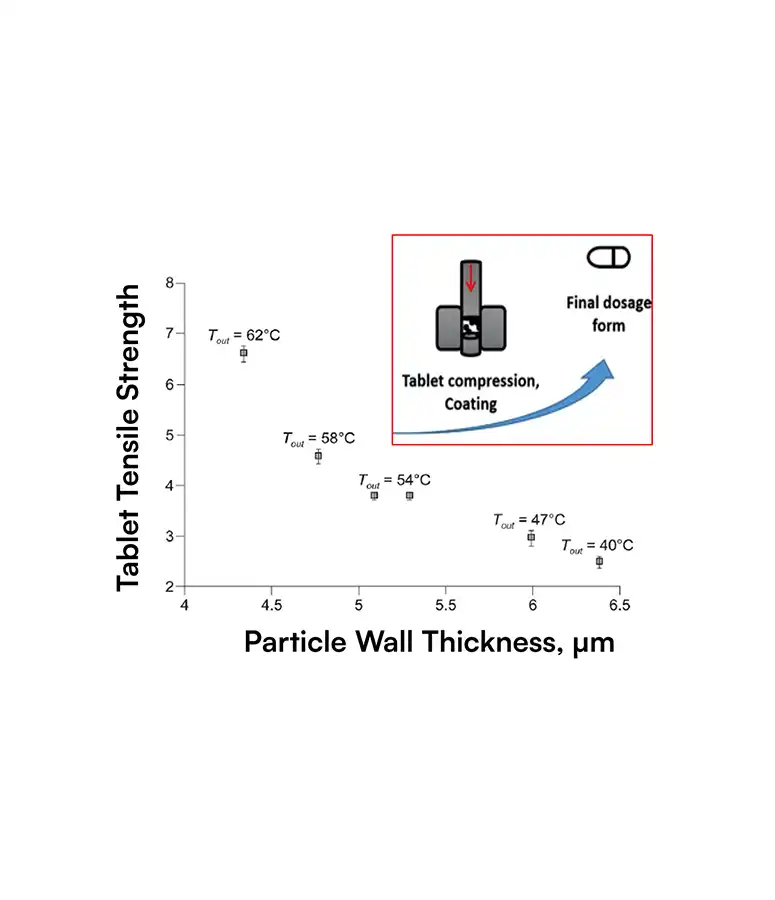
SDD particles have complex, hollow morphologies that strongly influence dissolution, stability, tablet strength, and ultimately bioavailability and yield.
Traditional tools cannot quantify these structures at scale, so teams only see the business impact as variability: failed dissolution tests, tablet defects, site‑to‑site differences, clinical surprises.
Business Outcomes
Our Solution: Microstructure Insight Sprint
A defined 3 to 6 week engagment that combines 3D X-ray micro CT imaging and advanced AI analytics to:
What You Get
Formulations
Studied

Transform Your Program with Microstructure Science
Get started with a drug product digital twin.