Injection Rate Impact on Long-acting Injectable Depot Dissolution in Ex Vivo Porcine Tissue
Comparing the performance attributes of an on-body injector and autoinjectors

Understanding the impact of device injection rate on depot release kinetics
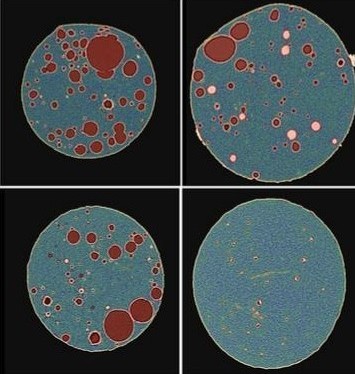
In collaboration with our partners from Gilead, this project examined how different injection rates affect the depot morphology and release kinetics of a high viscocity formulation. Of particular interest was the comparison of an on-body injection (OBI) system for self admistration with traditional injectors, including a slow auto-injector (AI), a fast auto-injector, and a fast universal tester (UT).

To assess this, X-ray micro-CT was used image ex vivo porcine tissue containing depots injected with each device. Through digital analysis, critical depot properties including volume, surface area, size, roundness, and tissue localization were measured. Finally, image-based release prediction was used to predict drug release profiles directly from the digitized depots' real morphology and shape attributes.

The study found significant differences across each device, the OBI producing a rounder depot, while traditional injectors tended to be flatter. A rank order difference in surace area to volume raito was also identified. For some devices, the depot was found to intrude into the intramuscular space, suggesting device type may impect permeation of the depot and localization into unpreferential tissue layers. Through the predicted release profiles, the depot from the OBI injection was found to have siginificantly longer release. Depots from slow AI, fast AI, and fast UT had faster release, following a rank order with good correspondance to their surface area to volume ratios.
Additional Case Studies
Our Expertise
In Numbers
Programs Supported
Formulations Digitized
Pharmaceutical Partners

Transform Your Program with Microstructure Science
Get started with a drug product digital twin.