Lyo Controlled Ice Nucleation
Assessing Process Performance of Controlled Ice Nucleation

Analyzing the controlled ice nucleation process with a focus on the process performance and product attributes
What is controlled ice nucleation?
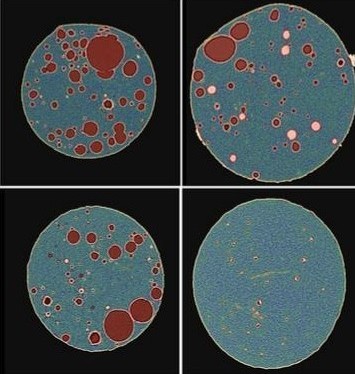
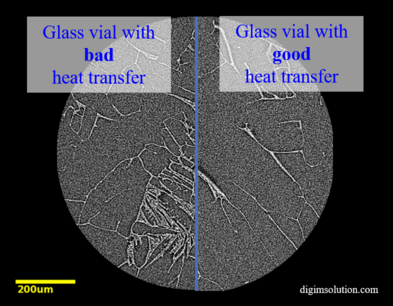
Lyophilization, or freeze-drying, has been around for centuries, dating back as far as the 13th century. However, in the past few decades there has been little change to process design, monitoring, and quality control assessment. Controlled ice nucleation (CIN) technology is a new method to improve efficiency in time and cost for lyophilization in addition to increasing batch homogeneity and product quality.

The technique involves cooling the batch to a specific temperature in order to reduce the randomness that ice generation can bring to a lyophilized sample such as ice crystal size and distribution. DigiM’s non-invasive X-ray imaging has been used to improve the understanding of the relationships between CIN process parameters and performance with product quality attributes. With AI technology, microstructure quality attributes including microporosity and cake wall thickness can be directly quantified.
Additional Case Studies
Our Expertise
In Numbers
Programs Supported
Formulations Digitized
Pharmaceutical Partners

Transform Your Program with Microstructure Science
Get started with a drug product digital twin.